|
You're at that time in your life - wise and with the evidence to prove it. We have invented technology that takes us to Mars but we are still struggling to come up with a solution for gray hair. Unless it's the Grecian Formula or Restoria Discreet. I had never heard of these products until someone I know mentioned that he was using Restoria and really liked how his hair was - stayed in place and made his grays look cool. Of course I was intrigued and immediately looked up the ingredients: Restoria Discreet color restoring cream: Water (Aqua), Glycerin, Alcohol Denat., Sulphur, Lead Acetate, Hydroxypropyl Methylcellulose, Lauramide DEA, Fragrance (Parfum), Denatonium Benzoate Lead acetate is is the key ingredient here: 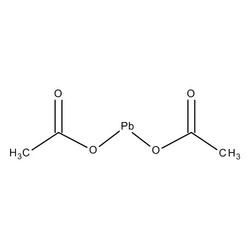 How does it work? This is how Grecian Formula describes: "Grecian works with the hair's natural chemistry to produce a similar-acting pigment where the melanin used to be. It combines with the hair's protein inside the hair to gradually "reverse" the graying process and bring back natural looking color. Grecian works for any color hair because you control the process so you determine when you regain your right color. You can even keep a little gray around the sides if you like." Here's how it actually works: lead reacts with oxygen in the air and sulfur in hair protein (keratin) to form oxides and sulfides which are dark. So how safe is this product? 1) The FDA considers lead acetate to be safe if appropriate warnings are posted on the product box: "The safety data submitted in support of this petition included results from trials on humans using the products. In the trials, people using the product under controlled conditions of use were monitored for the amount of lead in their bloodstream. No significant increase in blood levels of lead was seen in the trial subjects and the lead was not shown to be absorbed into the body through such use." The ways of the FDA are mysterious - lead acetate is banned in Europe and Canada because of toxicity concerns. According to this review on lead toxicity, there is NO safe level of exposure to lead. 2) The concentration of lead acetate i hair products like Grecian Formula and Restoria is twenty times that found in lead paint: Lead acetate is also very soluble in water. So imagine this: you apply the cream on your head, forget to wash your hands. You touch stuff, your child's toy that she will stick in her mouth, eat a bagel. Or you have dandruff, scratch your head and get a small lesion on your scalp - now a conduit for this poison to get into your body. Just not worth it. In fact, these products have so much lead you can't just throw them away - they need to be disposed as hazardous waste.
Other resources to enhance awareness on toxicity of everyday products, including hair dyes: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2700586/ Where lead hides: http://hydra.usc.edu/scehsc/pdfs/D-1-3-2%20Where%20Lead%20Hides.pdf So what can you do? Read the ingredients and get educated. Throw (or dispose appropriately) toxic products. For your grays? Embrace them. In the next post we'll look at some research on using natural ingredients to slow graying and prevent hair loss. Stay tuned!
2 Comments
Photo credit: http://lethow.com/home-remedies/body-odor-removal/ Now that we understand what causes body odor, it's time to talk about how to prevent it. I am not comfortable about plugging sweat glands - sweat cools the body and is a very natural phenomenon that I am not looking to suppress. So now it was about getting effective ingredients to kill the bacteria that produce body odor. I tried several experiments. 1) Baking soda: Or sodium bicarbonate, the ubiquitous household chemical. Baking soda kills bacteria and keeps you dry. The most common DIY deodorant recipes on the internet use baking soda as the key ingredient. Pros: Effective! I made a pretty good deodorant stick using baking soda, arrowroot powder, coconut oil, and cocoa butter. I could walk an hour in scorching heat and still smell pretty good at the end of the day. BUT Cons: After weeks of use, my pits were on fire! Sodium bicarbonate has a high pH - i.e. it is an alkaline substance. Our skin likes to be slightly acidic. Second, the stick stained the sleeves of my tops a yellow color - I was not a fan of that. Third, the physical condition of this stick depended heavily on the climate - what was a perfectly nice and smooth stick i California melted into a gooey mess when I took it with me to India. 2) Acetic acid or vinegar: Vinegar is an acid and the pH prevents bacteria from thriving. Of course it smells a bit like body odor so you need to add things to it even after diluting it heavily for it to do anything. Pros: Not irritating to skin. Cons: At the dilution I was comfortable with, it was not very effective. Lasted only half a day and if I took one of my walks, I could smell myself after. 3) Magnesium oil aka Magnesium chloride: Magnesium is an element needed for our tissues to be healthy. While I was doing my research, this was a reasonably common DIY recipe for body odor. You make up a solution of magnesium chloride, add some essential oils, and voila! Pros: Not irritating, and your body gets a healthy dose of magnesium! Cons: Again like vinegar, not very effective. Gave me protection for only half the day. I tried other recipes with witch hazel and zinc oxide, etc. etc. And then I read a bunch of papers and learned that: 1) Magnesium chloride showed significant anti-microbial activity at low pH 2) some essential oils are better than others for anti-microbial activity 3) zinc oxide is effective for body odor due to its reaction with short chain fatty acids such as isovaleric acid, that are produced when bacteria act on sweat like we saw in this post. Although Zn is found in our cells and is needed for our bodies to function healthily, the non-nano zinc oxide particles are too large to enter our bodies. So I put all this together into a spray formulation - water, vinegar, magnesium chloride, essential oils of lemon, tea tree, and lavender and zinc oxide. Since I am using non-nano zinc oxide, I believe the antibacterial effect will be diminished. So how did this work out? All day, with a 40 minute brisk walk in the sun - smell fine. I can still smell some of the essential oils! It has exceeded my expectations. Plus the goodness of magnesium - a bonus. A couple samples will be sent out to some people for feedback. For now, me and my daughter are sticking to this. I've been trying to make a deodorant for some time now. My tween has officially joined the body odor group :( and I have been attempting to make something strong enough to last through PE and dance. My goal was to make something that would control the body odor using ingredients healthy enough to eat or at the very least, non-toxic. In this post series, I share my thought process as I formulate such a deodorant. But first, we need to talk about the biochemistry of body odor. Yay! Body odor is caused by the breakdown of sweat into foul smelling acids by some of the harmless bacteria our bodies house. Sweat itself is odorless. Propionibacteria live in the ducts of the sebaceous glands of adult and adolescent humans and break down sweat into propionic acid. Staphylococcus epidermidis breaks down sweat into isovaleric acid. To prevent body odor, the cosmetics industry has gone the route of "no sweat, no stink" aka the antiperspirant. Antiperspirants contain an aluminum or aluminum zirconium salts that works to plug the sweat glands and thus prevent sweat from reaching the skin's surfact. The the higher the concentration of the salt, the better it works. Basically the pH of sweat causes the aluminum to precipitate and thus block the sweat gland. There have been associations of aluminum with cancer and Alzheimer's disease. While there might be a connection between the element and these diseases, it is unlikely that they are caused by the use of antiperspirants. This really long paper is an excellent summary of aluminum, toxicity: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2782734/www.ncbi.nlm.nih.gov/pmc/articles/PMC2782734/ A less common and less effective product that has been used for body odor is the deodorant. Deodorants work by killing the bacteria that cause those smelly acids to form. Typically, deodorants contain chemicals like alcohol, propylene glycol, and sodium bicarbonate (baking soda) as key ingredients. The problem with these ingredients is that they are skin irritants. Often times, deodorants and antiperspirants are used interchangeably - many aluminum containing products are labeled as deodorants: Now that we have covered the basics, stay tuned for part 2 where I explore formulating a deodorant using ingredients that are naturally found in our bodies or derived from plants. Why do I care? For one, why should we use products blindly without caring about what we put on our bodies? I want to be empowered to use products that I will not regret using. As a mother I feel compelled to make good choices for my kids in all respects. Second, nature always has a solution. However, these are not well publicized. In part 2, I will go through scientific literature (published in peer reviewed scientific journals - I love PubMed! - http://www.ncbi.nlm.nih.gov/pubmed) to formulate a deodorant.
|
|
|
© Saroya Natural 2023 | All rights reserved.
Website designed by Aadi M |
Contact Us |





 RSS Feed
RSS Feed